PEATMOSS TOPDRESSING CONTROL OF TAKE-ALL
ROOT ROT
ON ST. AUGUSTINE GRASS
Phillip F. Colbaugh, Xikui Wei. and l. A. McAfe
Texas A&M Research Center at Dallas April, 2003
INTRODUCTION
In recent years we have discovered that underground organs of turf
grasses are commonly attacked by ectotrophic fungi that cause
destructive patch diseases. Ectotrophic fungi grow over living
turf grass roots and underground stems as runner hyphae (dark
fungal threads). There are several ectotrophic fungi that cause
turf grass diseases and their appearance is similar on the different
turf grass hosts they attack. For convenience, all of these fungi
are referred to as ETRIF (ectotrophic root infecting fungi) to
simplify their diagnosis and associations with the similar turf
diseases they cause.
Take-all root rot (TARR) of St Augustine grass has emerged as
a major problem on landscapes in Texas as well as other states along the Gulf Coast
including Florida. The disease is caused by Gaeumannomyces graminis
var. graminis, which belongs to the ETRIF pathogen group. The
brown-black mycelial growth of the fungus (Fig.1) colonizes roots,
stolons and shoots but it is primarily a root destroying pathogen.
Damaging effects of this disease on St. Augustine grass were first
observed and described in Texas by Dr. Joseph Krausz (plant pathologist
at Texas A&M University) and in Florida by Dr. Monica Elliott
(University of Florida). In a 1999 survey of St. Augustine grass
lawns in north Dallas, we observed yellow patch symptoms (Fig.
3) of the disease on 61% of 70 lawns during the month of September.
If this disease progresses it kills the stolons and produces patches
of dead grass during summer ranging from 3-10 ft in diameter.
Because of the widespread nature of this disease, our research
investigations sought to develop a practical control measure for
landscapes with St. Augustine grass lawns.
landscapes in Texas as well as other states along the Gulf Coast
including Florida. The disease is caused by Gaeumannomyces graminis
var. graminis, which belongs to the ETRIF pathogen group. The
brown-black mycelial growth of the fungus (Fig.1) colonizes roots,
stolons and shoots but it is primarily a root destroying pathogen.
Damaging effects of this disease on St. Augustine grass were first
observed and described in Texas by Dr. Joseph Krausz (plant pathologist
at Texas A&M University) and in Florida by Dr. Monica Elliott
(University of Florida). In a 1999 survey of St. Augustine grass
lawns in north Dallas, we observed yellow patch symptoms (Fig.
3) of the disease on 61% of 70 lawns during the month of September.
If this disease progresses it kills the stolons and produces patches
of dead grass during summer ranging from 3-10 ft in diameter.
Because of the widespread nature of this disease, our research
investigations sought to develop a practical control measure for
landscapes with St. Augustine grass lawns.
DESCRIPTION OF FIELD SYMPTOMS
Symptoms of take-all root rot disease (TARR) typically appear
on St Augustine grass as diseased patches of turf during late
spring and throughout the summer months. Pathogen activity causes
a severe root rot that completely destroys tap roots which anchor
St. Augustine grass stolons to the ground. Visual symptoms of
the disease on lawns are initially small yellow patches of turf
with leaf blades that appear chlorotic while the healthy leaves
remain a typical green color (Fig. 3). The yellow patches are
thought to be associated with the production of a toxin by the
ETRIF fungus when the turf is growing under stressful conditions.
Yellows symptoms of the disease can persist on lawns throughout
the summer growing season. Dark brown or black mycelial threads
of this fungus (Fig. 1) are distinctive and produce scattered
black dots (hyphopodia) that anchor the fungus to the plant. Roots
of affected plants become shortened, discolored, and often have
dark colored lesions that are visible upon inspection with a hand
lens. Eventually the roots become completely rotted and shriveled
to form a non-functional root system (Fig 2). In the final stages
of decline, diseased stolons gradually succumb to hot summer temperatures
or cold winter weather and produce large patches of dead grass
that do not recover from injury.
Affected patches of turf can at first be quite small ranging
from 1-2 feet in diameter; however, they also appear as larger
areas that can range from 5-10 feet in diameter. Diseased areas
are not always circular but often appear as roughly circular patterns
in the lawn. In our 2002 TARR survey on North Dallas lawns, we
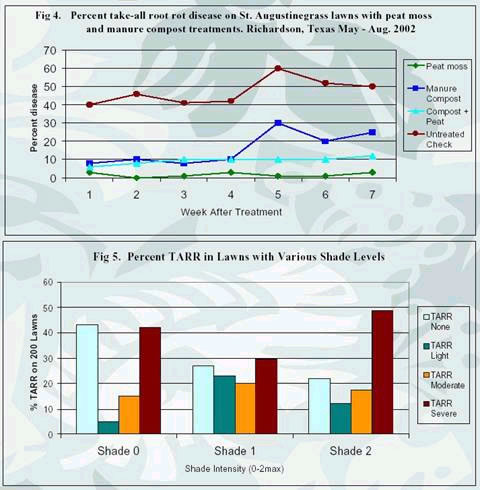
observed a higher number of take-all symptoms in heavily shaded
areas compared to areas receiving direct sunlight or partial shade
for most of the day (Fig. 5). TARR disease should not be confused
with white grub damage which can also appear at the same time
of the year. The best clue is to look for the yellow or chlorotic
leaf extensions (fig. 3) on St. Augustine grass turf that has
not received mowing for several days. Symptoms of TARR disease
also include the appearance of brown shriveled roots that are
killed by the fungus as opposed to white grub damage where the
roots are actually removed by insect feeding.
SEARCH FOR A PRACTICAL DISEASE CONTROL ON DALLAS HOMELAWNS
We used two approaches to control the TARR disease in field investigations
on area lawns during the past three years.
One approach utilized conventional fungicide sprays with Terraguard
®, Bayleton®, Heritage®, and Banner Maxx® using
2.9L of spray per 10 m2. A second approach utilized topdressing
lawn care products including (1) manure compost and (2) sphagnum
peat moss. Manure products can enrich the microbial number and
diversity for variable lengths of time and low pH products like
peat moss had been shown to suppress the Gaeumannomyces fungus
in previous research. While some of the manure based topdressing
regimens demonstrated improved turf grass growth, effects on disease
control were only partial and limited in duration. Research field
plots with the fungicides Terraguard ® (4 - 8 oz) or Bayleton®
(2 oz) treatments gave good results for controlling the take-all
root rot symptoms. Success with fungicide treatments was better
on a lawns maintained under shaded conditions compared to lawns
in full sunlight.
A second approach with topdressings used low pH topdressing with
sphagnum peat moss. This topdressing approach has consistently
demonstrated TARR disease suppression in field studies during
the past two years. Our field comparisons of manure compost vs.
peat moss topdressings indicate the peat moss to be a more effective
long-term approach for reducing symptoms of the TARR disease.
Some of the older research literature on the fungus causing TAP
indicates its aversion to low pH. This might explain how the peat
moss (pH = 4.4) controls the fungus on exposed stolons and roots
where the disease is active.
CONCLUSIONS
There is no indication of varietal resistance to take-all root
rot since the disease has been noted on all of the commercial
St. Augustine grass varieties. The use of fungicide applications
is also limited with only a few fungicides that are approved for
use on this disease. Although there is good evidence that fungicides
are capable of controlling the disease, environmental conditions
and vigor of the turf may pose some limitations on the effectiveness
of fungicide treatments. At this time we have no explanation as
to why we observed a lack of uniformity in fungicide effectiveness
on different lawns.
The use of organic topdressing to control turf grass disease
is a relatively new approach to controlling turf grass diseases.
Because of the complexity of microbial antagonism, fertility values
of topdressing materials, different types of diseases and susceptibility
of pathogens to pH, most of this type of research is directed
by trial and error experimentation. We do have good evidence that
the acid peat moss topdressings result in control of TARR on St.
Augustine grass on Dallas area home lawns. In comparison studies,
peat moss topdressing reduced symptoms of TARR for longer periods
than cow manure compost and is thus considered the more effective
disease control product. Additional research will address the
best time to apply peat moss topdressing products as well as possible
effects on other turf grass pathogens and diseases.

Fig. l. Mycelial growth on stolon

Fig. 2. Take all root rot symptoms on St. Augustine grass

Fig. 3. Yellow patch symptoms
Gentlemen, we have done the hard calculations on use of peatmoss on urban lawns. You wanted to know how much peat to put down. After exhaustive field testing we figure we are using just about the equivalent of one 3.8 cubic foot bale peat per 1000 square feet of turf.
One bale per 1000 square feet is a good figure to use if someone needs to know. Thanks, Phil
|



